 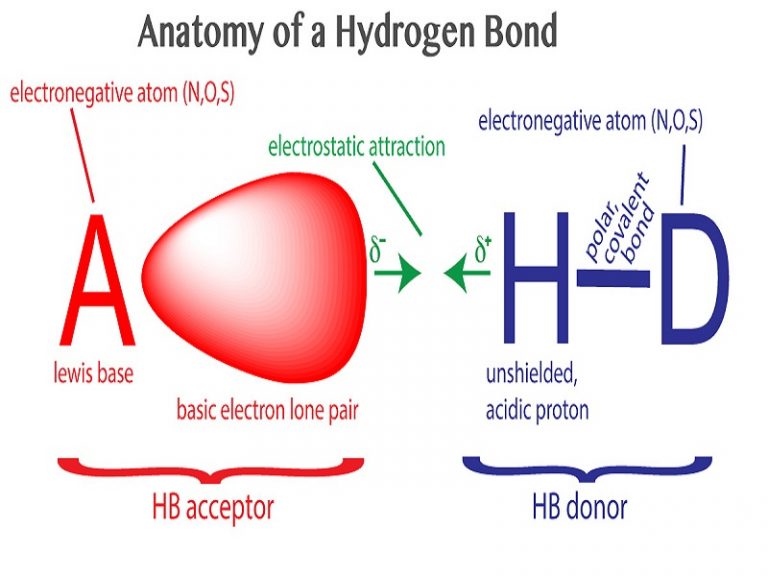
This means the molecules will be soluble in a polar solvent such as water. The presence of hydrogen bonding between molecules of a substance indicates that the molecules are polar. Molecules which are capable of hydrogen bonds have hydrogen atoms which are covalently bonded to highly electronegative elements (O, N, F). In summary, hydrogen bonds are (relatively weak) intermolecular forces, while covalent and ionic bonds are (relatively strong) intramolecular forces. Hydrogen bonds have strengths ranging from 5 kJ/mol to 50 kJ/mol. Water is the best-known compound that has hydrogen bonds. They exist when you have a negative #"O"#, #"N"#, or #"F"# atom in one molecule and a positive #"H"# atom attached to an #"O"#, #"N"#, or #"F"# atom in another molecule. Hydrogen bonds are especially strong intermolecular forces. The covalent bonds are intramolecular bonds because they hold the atoms together in a single molecule.Ĭovalent bond strengths range from 100 kJ/mol to 1100 kJ/mol. Ionic bond strengths (lattice energies) range from 600 kJ/mol to 6000 kJ/mol.Ĭovalent bonds form when two atoms share electrons. Ionic bonds are intramolecular bonds, because the ions exert forces among the ions of the same compound. The ions in ionic solids are close to each other, so ionic attractions are strong. The electrostatic attraction between these ions is an ionic bond. The atom that gains an electron becomes a negative ion. The atom that loses an electron becomes a positive ion. Ionic bonds form when one atom transfers electrons to another atom. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
